What is the result of distilling a mixture of 80% $\\text{HN}{{\\text{O}}_{3}}$ and 20% ${{\\text{H}}_{2}}\\text{O}$?\n \n \n \n \n A. Pure ${{\\text{H}}_{2}}\\text{O}$ and azeotropic mixture can be separated.B. Pure ${{\\text{H}}_{2}}\\text{O}$ and
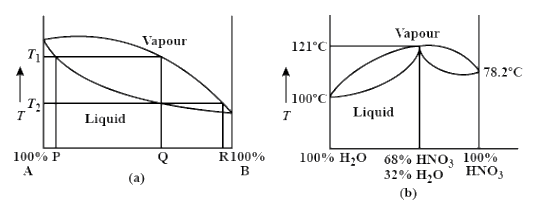
What is the result of distilling a mixture of 80% $\\text{HN}{{\\text{O}}_{3}}$ and 20% ${{\\text{H}}_{2}}\\text{O}$?\n \n \n \n \n A. Pure ${{\\text{H}}_{2}}\\text{O}$ and azeotropic mixture can be separated.B. Pure ${{\\text{H}}_{2}}\\
PEPTIDES New Drug Approvals

Financial Modeling Using Quantum Computing (2023), PDF, Quantum Computing

Item ID Number D Corporate Author Year Month/Day Color Number of

What is the result of distilling a mixture of 80% $\text{HN

SOLVED: Q.15.0 Marks: A binary mixture of n-heptane and n-octane

FR2795726A1 - NEW FUNGICIDE PYRAZOLES - Google Patents

PEPTIDES New Drug Approvals

CHEM 282 Virtual Lab 5 - Carran. - CHEM 282 Virtual Experiment #5: Simple and Fractional - Studocu

Solved Procedure Simple distillation: 1. Place about 30 mL

Solved 1. The following data was obtained when a sample

Consider the following curve :What iss the result of distilling a

CHEM 282 Virtual Lab 5 - Carran. - CHEM 282 Virtual Experiment #5